Japanese Doctors Perform World's First Living Donor Lung Transplant on COVID-19 Patient
Doctor's at Kyoto University Hospital have performed the world's first living donor lung transplant on a COVID-19 patient.
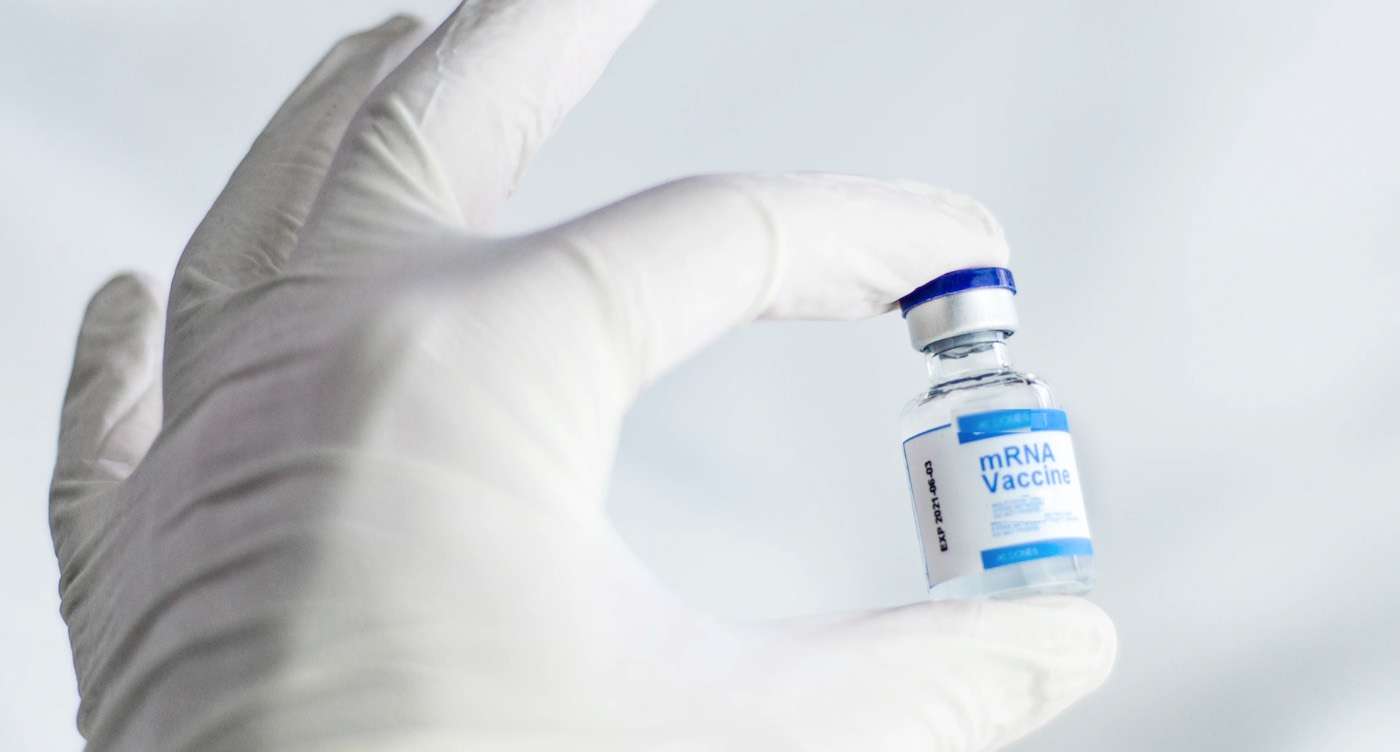
A phase 1 clinical trial of an experimental vaccine primed the immune system using a unique approach in order to prevent HIV.
The promising results, announced in February by the International AIDS Vaccine Initiative (IAVI) and Scripps Research, the vaccine showed success in stimulating production of rare immune cells needed to generate antibodies against the fast-mutating virus—and the targeted response was detected in 97 percent of participants who received the vaccine.
"We showed that vaccines can be designed to stimulate rare immune cells with specific properties, and this targeted stimulation can be very efficient in humans," said William Schief, PhD, a professor and immunologist at Scripps Research and executive director of vaccine design at IAVI's Neutralizing Antibody Center, whose lab developed the vaccine.
"We believe this approach will be key to making an HIV vaccine and possibly important for making vaccines against other pathogens."
As a next step, IAVI and Scripps are partnering with the biotechnology company Moderna to develop and test an mRNA-based vaccine that harnesses the approach to produce the same beneficial immune cells. Using mRNA technology could significantly accelerate the pace of HIV vaccine development.
HIV, which affects more than 38 million people globally, is known to be among the most difficult viruses to target with a vaccine, in large part because it constantly evolves into different strains to evade the immune system.
"These exciting findings emerge from remarkably creative, innovative science and are a testament to the research team's talent, dedication and collaborative spirit, and to the generosity of the trial participants," says Mark Feinberg, MD, PhD, president and CEO of IAVI.
For decades now, HIV researchers have pursued the holy grail of stimulating the immune system to create rare but powerful antibodies that can neutralize diverse strains of HIV. Known as "broadly neutralizing antibodies," or bnAbs, these specialized blood proteins could attach to HIV spikes, proteins on the virion surface that allow the virus to enter human cells, and disable them via important yet difficult-to-access regions that don't vary much from strain to strain.
"We and others postulated many years ago that in order to induce bnAbs, you must start the process by triggering the right B cells—cells that have special properties giving them potential to develop into bnAb-secreting cells," Schief says.
"In this trial, the targeted cells were only about one in a million of all naïve B cells. To get the right antibody response, we first need to prime the right B cells. The data from this trial affirms the ability of the vaccine immunogen to do this."
The priming step would be the first stage of a multi-step vaccine regimen aimed at eliciting many different types of bnAbs, he says.
The strategy of targeting naïve B cells with specific properties is called "germline-targeting," as these young B cells display antibodies encoded by unmutated, or "germline" genes. Researchers believe the approach could also be applied to vaccines for other challenging pathogens such as influenza, dengue, Zika, hepatitis C viruses and malaria.
"This is a tremendous achievement for vaccine science as a whole," says Dennis Burton, PhD, professor and chair of the Department of Immunology and Microbiology at Scripps Research, scientific director of the IAVI Neutralizing Antibody Center and director of the NIH Consortium for HIV/AIDS Vaccine Development. "This clinical trial has shown that we can drive immune responses in predictable ways to make new and better vaccines, and not just for HIV. We believe this type of vaccine engineering can be applied more broadly, bringing about a new day in vaccinology."
The clinical trial, IAVI G001, took place at two sites: George Washington University in Washington, D.C., and the Fred Hutchinson Cancer Research Center in Seattle, enrolling 48 healthy adult volunteers. Participants received either a placebo or two doses of the vaccine compound, eOD-GT8 60mer, along with an adjuvant developed by the pharmaceutical company GSK.
"This is a landmark study in the HIV vaccine field, demonstrating success in the first step of a pathway to induce broad neutralizing antibodies against HIV-1," McElrath says.
The study sets the stage for additional clinical trials that will seek to refine and extend the approach—with the long-term goal of creating a safe and effective HIV vaccine.
Funding from the Bill & Melinda Gates Foundation, through the Collaboration for AIDS Vaccine Discovery—and grants from the National Institute of Allergy and Infectious Diseases (NIAID)—supported a network of partners conducting analyses and contributing to the vaccine development, including the NIH Vaccine Research Center, the government of the Netherlands, and the US Agency for International Development.
(SOURCE: Scripps.edu)
SHOOT This HIV News to Your Global Audience on Social Media…
Be the first to comment